The Pharmaceutical Care Management Association (PCMA) is the national association representing America’s pharmacy benefit managers (PBMs). PBMs administer prescription drug plans for more than 266 million Americans who have health insurance from a variety of sponsors including: commercial health plans, self-insured employer plans, union plans, Medicare Part D plans, the Federal Employees Health Benefits Program (FEHBP), state government employee plans, managed Medicaid plans, and others.
PCMA continues to lead the effort in promoting PBMs and the proven tools they utilize, which are recognized by consumers, employers, policymakers, and others as key drivers in lowering prescription drug costs and increasing access.
PBMs are projected to save employers, unions, government programs, and consumers $654 billion – up to 30 percent – on drug benefit costs over the next decade, according to research from Visante.
PBMs reduce drug costs by:
- Offering Amazon-style home delivery of medications and creating select networks of more affordable pharmacies;
- Encouraging the use of generics and more affordable brand medications;
- Negotiating rebates from drug manufacturers and discounts from drugstores;
- Managing high-cost specialty medications;
- Reducing waste and improving adherence.
MEET THE REST OF OUR TEAM
Nunc quis fermentum tellus, id fringilla turpis. Pellentesque elit elit, placerat in lobortis volutpat, cursus sodales tortor. Ut tincidunt nulla justo, ut rhoncus mauris placerat vitae. Sed maximus massa nisi, sit amet viverra nunc efficitur id. Mauris luctus, arcu et elementum pulvinar, odio enim pharetra ex.
Phasellus diam magna, mollis eget justo sit amet, tincidunt sagittis lorem. Donec vestibulum augue erat, a convallis nisi gravida eu. Pellentesque luctus sem ex, non varius libero efficitur eget.
Meet the TeamPBMs: Committed to Helping Patients
The Critical Path Forward: Rx
Policies to Reduce Patient Costs,
Improve Access
For America’s pharmacy benefit managers, PBMs, our core mission is to increase access to affordable prescription drugs for all Americans.
But more can be done and we stand ready to be part of the solution.
PCMA’s Policy Platform, outlined below, presents straightforward solutions that, taken together, would lower prescription drug costs and make pharmaceutical care even more accessible for all Americans.
This policy platform would result in total Federal Savings of $257.5 billion to $398.7 billion over ten years.
QUICK LINKS
PART ONE
Modernize Medicare Part D
PART TWO
End Anticompetitive
Practices and Enhance
Competition
PART THREE
Build a Value-driven,
Equitable Health Care
System
PART ONE
Modernize Medicare Part D
In its first 15 years, Medicare Part D has significantly improved the affordability of prescription drugs for people with Medicare.
PBMs support a set of proposals, which, taken together, would modernize Part D and better align the incentives of all stakeholders to control drug costs and improve quality and outcomes for beneficiaries. These include policies to:
A) Improve Affordability
B) End Misaligned Incentives
C) Increase Choice and Competition
D) Keep Premiums Affordable
E) Achieve The Triple Aim
Modernize Medicare Part D
Total Federal Savings:
$280.3 billion over
ten years.
A) Improve Affordability
Unlike most patients with commercial drug coverage, Part D beneficiaries can face unexpected, high costs because
the Part D drug benefit does not have an annual limit on out-of-pocket spending.
We should cap annual out-of-pocket costs to provide better financial protection for beneficiaries with high drug costs
and make cost-sharing more affordable and manageable.
B) End Misaligned Incentives
Rather than lowering the increasingly high prices they set for their drugs, manufacturers want to shrink the discounts they owe Part D beneficiaries and taxpayers.
Instead, to lower drug prices and reduce costs, we should end misaligned incentives and hold manufacturers accountable for their high prices and require manufacturer contributions throughout the Part D benefit phase—from Initial Coverage to Catastrophic.*
*We believe that manufacturer responsibility should be higher in the Catastrophic Coverage phase and particularly for protected-class drugs and novel drugs without therapeutic competition and very high costs.

C) Increase Choice and Competition
The most important barrier to bringing costs down is the lack of competition and rules that stymie beneficiary choice and competition. By increasing competition, we can lower costs, increase access, and drive continued innovation in Part D.
We should build on Part D’s record of success by eliminating the two drugs per-class requirement; allowing select exclusions of the protected classes; encouraging beneficiaries to use lower cost drugs, such as generics and biosimilars; and promoting greater plan choices for beneficiaries.

D) Keep Premiums Affordable
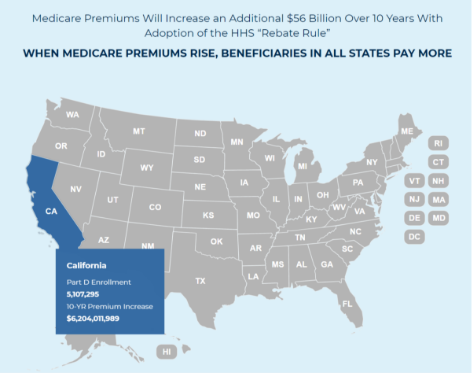
The rebate rule, which limits plans and PBMs from negotiating lower drug costs on behalf of Part D beneficiaries, would increase premiums 25% and cost taxpayers $170 billion.
A 2019 Government Accountability Office report found that PBMs passed 99.6% of all rebates on to plan sponsors, helping keep premiums and costs low. We should repeal the “rebate rule” and advance real solutions to bring down drug costs.
E) Achieve the Triple Aim
We should advance transformation in the delivery of pharmacy care services by rewarding value-driven pharmacies that promote quality care, improve clinical outcomes, and reduce out-of-pocket costs for Part D beneficiaries.
Rather than unravel value-driven gains, we can improve pharmacy pay-for-performance (“DIR”) by ensuring pharmacies know how they’re being measured, including through tools like EQuIPP, and requiring Pharmacy Administrative Service Organizations (PSAOs) to share with pharmacies the quality measures and criteria that they are required to meet.
PART TWO
End Anticompetitive Practices and
Enhance Competition
Increased competition in the marketplace is essential for PBMs to effectively lower drug costs.
But brand drug manufacturer tactics that block generics and biosimilars from entering the market weaken PBMs’ ability to negotiate and deliver lower prescription drug costs. These pricing strategies, including “patent thickets,” “evergreening” and “product hopping,” unfairly protect some prescription drugs and biologics from more generic and biosimilar competition.
The result? Higher costs for patients and payers. We can do better by eliminating patent thickets, limiting evergreening and product hopping, and clearing the way for other manufacturers to bring their products to market, paving the way for more competition and lower costs.
End Anticompetitive Practices and
Enhance Competition
Total Federal Savings:
Currently there are
only
29
Qbiosimilars on the
market in the
U.S. versus
80
Approved in Europe.
Reserve Market Exclusivities to True Innovation
Innovation in the prescription drug marketplace without affordability undermines patient access. Congress has granted over-long exclusivity periods for biologics and orphan indications leading to orphan-drug indication abuses and unforeseen delays in getting more affordable biosimilars to the market.
Reducing exclusivity periods for biologic medications and orphan drugs, along with reducing the first generic exclusivity period for certain non-first generics, will create more competition and lead to lower overall drug costs.
Ensure Drugs Can Compete Fairly
Drug manufacturers, which alone set and raise prescription drug prices, use a host of strategies for fending off fair competition. These range from shadow pricing to abusing the Food & Drug Administration’s (FDA’s) citizen petition process to marketing schemes that undermine the tools employers and other health plan sponsors use to keep costs down for everyone.
While holding manufacturers accountable for those prices, policymakers could enforce antitrust laws, reform the FDA’s citizen petition process, and outlaw coupons (where they are not already considered a kickback) for drugs with direct competition. Eliminating tax deductibility for direct-to-consumer advertising of prescription drugs would also reduce inappropriate demand for higher-cost drugs.
Promote Generic and Biosimilar Competition
The key to reducing prescription drug costs is through increased competition in the marketplace. As more biosimilars enter the market, increasing their uptake will help boost competition and lower costs for patients. This can be achieved by streamlining how new biosimilars come to market, educating physicians and patients on the efficacy of biosimilars, making biosimilars interchangeable, and requiring a common billing code to make the substitution process smoother.
In addition, modifying cost-sharing for beneficiaries receiving low-income subsidies in Medicare Part D would encourage greater use of generics, lower cost brands, and biosimilars.
PART THREE
Build a Value-driven, Equitable
Health Care System

Build a Value-Driven, Equitable Health Care System
Total Federal Savings:
PBMs are leading the effort to align reimbursement around the value of prescription drugs. Simply put, we should drive health care solutions that promote value and results, not volume.
PBMs and manufacturers negotiate value- and outcomes-based contracts for drugs that have quantifiable, widely-accepted outcomes. Data collected to inform these contracts continue to provide physicians and payers with insights that enhance clinical decision-making, improve patient health, and increase competition in the marketplace.
Accelerate Value-based Care
Greater adoption of value-based purchasing (VBP) and accelerating patient-focused pharmacy care can improve health outcomes. Providing Medicare Part D Prescription Drug Plans with access to Medicare Parts A and B claims data, establishing safe harbors for VBP contracting, and allowing Medicare Part D and state Medicaid plans greater flexibility to adopt private-sector formulary management techniques will provide a boost to these payment models.
Part D plans can utilize medical data in combination with prescription data to improve health outcomes. Legislative changes allowing the use of these data also would advance indication-based formularies and decrease prescriber burden.
Advance Use of Real-World Evidence
In a world where the cost of a given biologic can exceed an individual’s lifetime earnings, biopharmaceutical manufacturers should be expected to undertake ongoing research in their products, even after they are approved.
Health plans and PBMs need accurate, scientifically reliable information on prescription drugs throughout their lifecycles, from pre-approval for timely coverage decisions to post-market surveillance, and research into side effects and long-term efficacy for expedited approvals. These efforts should include rigorous evidence of drugs’ performance under real-world conditions.




